Chondrocyte
Chondrocytes are the only cells found in various types of cartilage (hyaline cartilage, fibrocartilage, and elastic cartilage), accounting for 1%–5% of the total cartilage volume. Three types of cartilage vary in the abundance, distribution, and types of collagens and proteoglycans, thus exhibiting different appearances and biomechanical properties.
Chondrocytes are metabolically active to maintain the renewal of the cartilaginous extracellular matrix (ECM) by synthesizing a significant amount of cartilage matrix components such as collagen, glycoproteins, proteoglycans (PGs), and hyaluronan [1,2].
What Is Chondrocyte Proliferation and Differentiation?
Chondrocytes originate from bone marrow mesenchymal stem cells (MSCs) during embryonic development through chondrogenesis. MSCs condense and proliferate into chondroprogenitors, which further differentiate into chondroblasts. Chondroblasts differentiate and mature into chondrocytes [3,4].
Chondrocytes eventually form the articular cartilage or enter the phase of endochondral ossification during which they experience proliferation, hypertrophy, and apoptosis, leading to the replacement of the hypertrophic cartilage with bone.
Chondrocyte proliferation and differentiation are tightly regulated by various factors, including key factors (such as Sox9, Runx2, and BMPs) and cell signals (such as Wnt, TGF-β, FGF, Ihh, and Nocth), extracellular matrix components (such as collagen, aggrecan), and mechanical stimuli. These factors act in concert to control the balance between chondrocyte proliferation and differentiation, ensuring proper cartilage homeostasis and function.
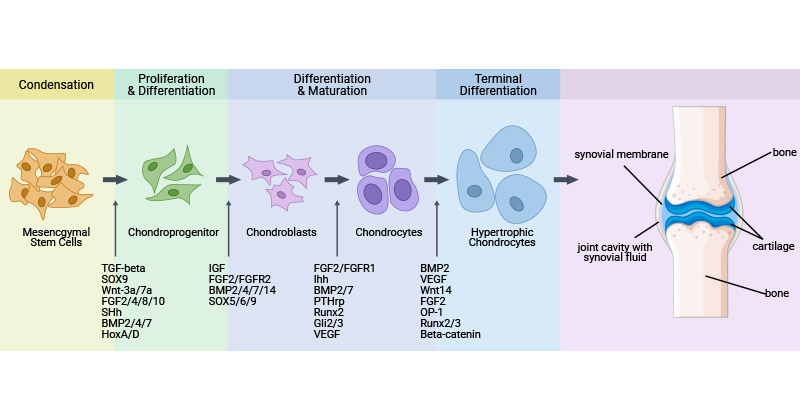
Figure 1. Chondrocyte proliferation and differentiation [4]
What Is the Structure of Chondrocyte?
Chondrocytes are embedded and clustered in the extracellular matrix (ECM) generated by themselves. Under the microscope, chondrocytes appear polygonal or round, with the exception that chondrocytes present in the tissue boundaries (e.g. the articular surfaces of joints) can be discoid or flattened.
Adult chondrocytes exhibit different morphologies in different layers due to their positions within cartilage. Cartilaginous superficial zone chondrocytes usually appear flattened and elliptical, aligned parallel to the surface. Chondrocytes in the mid-zone display a spheroidal shape with a larger size and are arranged randomly, whereas those in the deep zone are predominantly rounded and aligned in perpendicular columns.
Mature chondrocytes are mostly distributed in cartilage pits in groups of 2-8. Under the electron microscope, chondrocytes had protrusions and wrinkles, and there were a large number of rough endoplasmic reticulum, a developed Golgi complex, and a small number of mitochondria in the cytoplasm. Chondrocytes are buried in the cartilage interstitium. It is located in a small cavity called a cartilage lacuna (Figure 2).
Chondrocytes display phenotypic instability and cytoskeletal integrity can influence matrix metabolism, potentially affecting the resilience of cartilage.
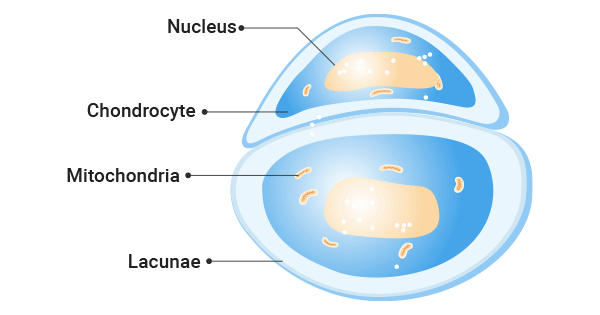
Figure 2. The Structure of Chondrocytes
What Is the Function of Chondrocyte?
Chondrocytes are the main cellular component of cartilage tissue and play crucial roles in the maintenance, repair, and adaptation of cartilage tissue, making them essential for joint health and function.
Chondrocytes synthesize the cartilage matrix and maintain cartilage homeostasis between the creation and destruction of extracellular matrix components [1,2]. Chondrocytes also respond to multiple biophysical cues, including transient thermal signals, oxygen tension, and mechanical stimuli.
Chondrocytes are essential for keeping homeostasis in the articular cartilage (AC) joints that act as cushions in joint movements, while chondrocytes of the growth plate regulate the growth of the epiphyseal plates [5].
Chondrocytes undergo hypertrophy and act as initial cartilage templates, followed by the invasion of blood vessels and osteoblasts, leading to the replacement of cartilage with bone during endochondral ossification [6]. They are useful for bone development.
Chondrocytes play a critical role in fracture healing by synthesizing cartilage, which serves as a foundation for endochondral ossification, stabilizing the healing fracture callus [7].
Overall, chondrocytes have multiple functions, including synthesizing ECM, regulating and maintaining cartilage homeostasis, responding to various biophysical cues, aiding in joint articulation, modulating the growth of epiphyseal plates, participating in fracture healing, and bone development.
Chondrocyte Markers
Chondrocytes express specific markers that distinguish them from other cell types. These markers are involved in chondrocyte maturation and cartilage formation (chondrocyte differentiation markers), maintaining the cartilage matrix, regulating chondrogenesis, and modulating chondrocyte function. Here list the most common markers of chondrocytes in the following table.
Chondrocyte and Disease Research
Chondrocytes, the sole cells found in cartilage, play a vital role in maintaining cartilage integrity and function. Abnormalities in chondrocyte behavior can lead to various diseases affecting cartilage and joint health.
Chondrocytes and Knee Arthritis
As mentioned before, chondrocytes play an irreplaceable role in the metabolism of cartilage. Running, jumping and other actions in daily life will cause the wear of joint cartilage. Knee arthritis is caused by the wear of articular cartilage. Articular cartilage is the most important structure in the joint. It plays a series of joint protection functions such as buffering stress and reducing friction. With the increase of age, repetitive sports injuries and changes in the body's conditions, the accumulated microtrauma and the accumulation of harmful substances in the joints will cause cartilage damage, cracking, and shedding, and eventually knee arthritis.
Chondrocytes and Osteoarthritis (OA)
In a healthy joint, chondrocytes maintain physiological and metabolic balance with strong redox control over differentiation and chondrogenesis [8,9]. A disruption in cartilage homeostasis leads to osteoarthritis. Chondrocytes in osteoarthritic cartilage transition to a hypertrophic phenotype and undergo apoptosis [10].
In the environment of a joint with osteoarthritis and under inflammatory conditions, chondrocytes undergo a metabolic imbalance and cartilage remodeling. This involves increased glycolysis, impaired mitochondrial function, and chondrocyte senescence [11,12].
Chondrocytes and Rheumatoid Arthritis (RA)
Chondrocytes of RA serve as both target and effector cells for these inflammatory mediators, leading to self-dysfunction and contributing to joint damage of RA [13]. In RA, pro-inflammatory molecules not only facilitate chondrocyte apoptosis but also disrupt chondrogenesis.
It is also evident that chondrocytes act as effector cells in RA directly and indirectly. Chondrocytes are directly involved in RA by releasing multiple enzymes of extracellular matrix degradation, promoting angiogenesis, and increasing inflammation and immune responses. Chondrocytes indirectly participate in RA through crosstalk with other relevant cells.
Understanding the molecular mechanisms underlying chondrocyte dysfunctions in disease states is crucial for developing targeted therapies. Researchers are exploring strategies to modulate chondrocyte metabolism, inhibit inflammatory pathways, and promote cartilage repair to prevent or slow down the progression of cartilage-related disorders.
References:
[1] Hemanth Akkiraju and Anja Nohe. Role of Chondrocytes in Cartilage Formation, Progression of Osteoarthritis and Cartilage Regeneration [J]. J Dev Biol. 2015;3(4):177-192.
[2] Archer CW, Francis-West P. The chondrocyte [J]. Int. J. Biochem. Cell Biol. 2003;35:401–404.
[3] Chen H, Tan XN, et al. Molecular Mechanisms of Chondrocyte Proliferation and Differentiation [J]. Front Cell Dev Biol. 2021 May 28;9:664168.
[4] Green JD, Tollemar V, et al. Multifaceted signaling regulators of chondrogenesis: Implications in cartilage regeneration and tissue engineering [J]. Genes Dis. 2015 Dec;2(4):307-327.
[5] Hunziker EB (1994). Mechanism of longitudinal bone growth and its regulation by growth plate chondrocytes [J]. Microscopy Research and Technique 28:505–519.
[6] Hinchliffe J.R., Gumpel-Pinot M. Control of maintenance and anteroposterior skeletal differentiation of the anterior mesenchyme of the chick wing bud by its posterior margin (the ZPA) [J]. J. Embryol. Exp. Morphol. 1981;62:63–82.
[7] Zhang C, Feinberg D, et al. Chondrocytes Promote Vascularization in Fracture Healing Through a FOXO1-Dependent Mechanism [J]. J Bone Miner Res. 2019 Mar;34(3):547-556.
[8] Y. Bai, X. Gong, C. Dou, Z. Cao, S. Dong. Redox control of chondrocyte differentiation and chondrogenesis [J]. Free Radic. Biol. Med., 132 (2019), pp. 83-89.
[9] R.K. June, R. Liu-Bryan, F. Long, T.M. Griffin. Emerging role of metabolic signaling in synovial joint remodeling and osteoarthritis [J]. J. Orthop. Res., 34 (2016), pp. 2048-2058.
[10] Linli Zheng, Ziji Zhang, Puyi Sheng a, Ali Mobasher. The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis [J]. Ageing Research Reviews Volume 66, March 2021, 101249.
[11] M. Cucchiarini, E.F. Terwilliger, D. Kohn, H. Madry. Remodelling of human osteoarthritic cartilage by FGF-2, alone or combined with Sox9 via rAAV gene transfer [J]. J. Cell. Mol. Med., 13 (2009), pp. 2476-2488.
[12] A. Mobasheri, M.P. Rayman, et al. The role of metabolism in the pathogenesis of osteoarthritis [J]. Nat. Rev. Rheumatol., 13 (2017), pp. 302-311.
[13] Tseng CC, Chen YJ, et al. Dual Role of Chondrocytes in Rheumatoid Arthritis: The Chicken and the Egg [J]. Int J Mol Sci. 2020 Feb 6;21(3):1071.


