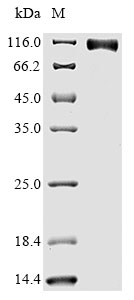
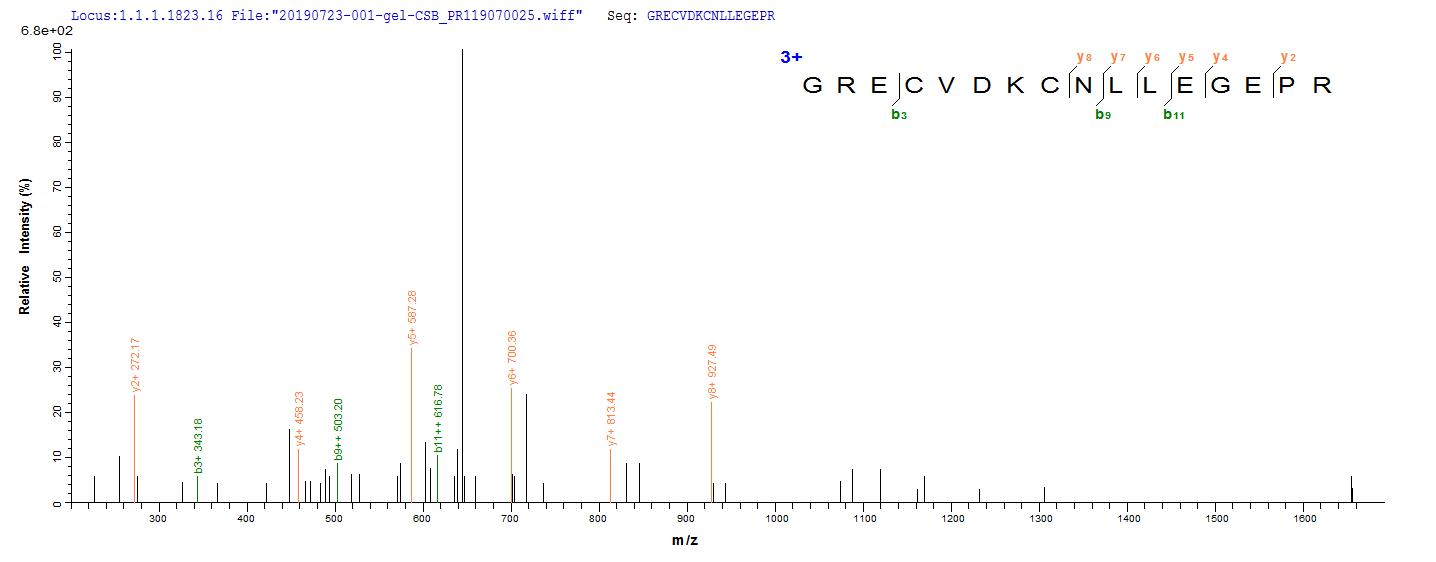
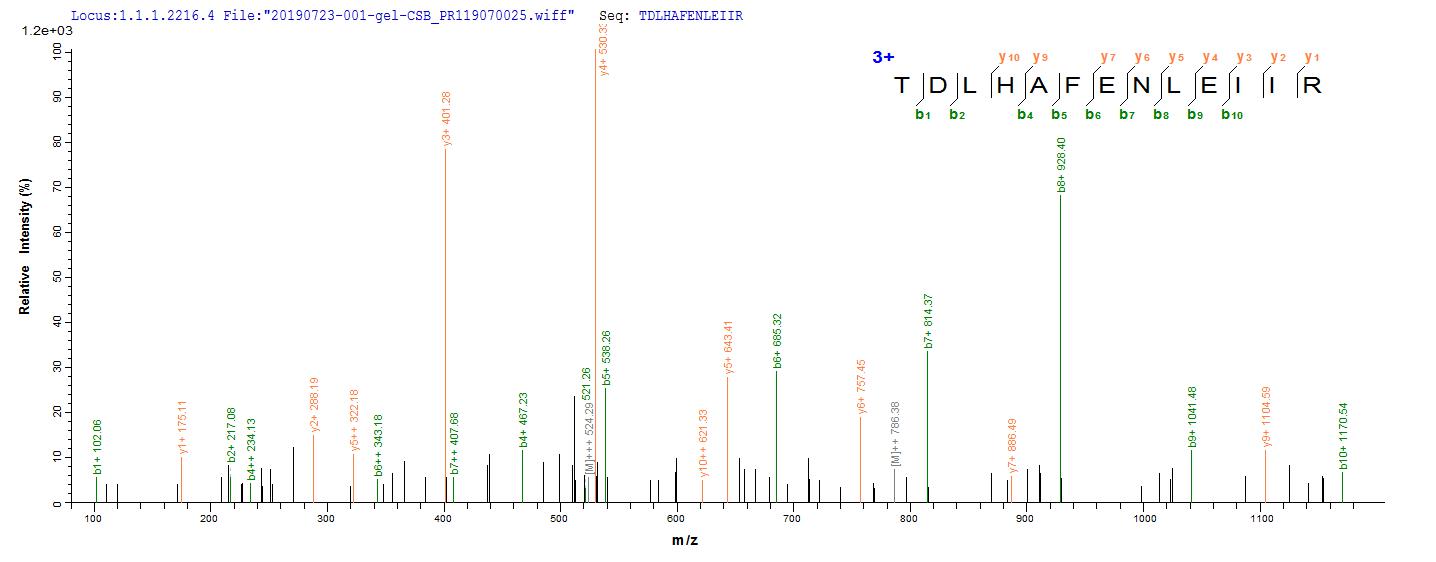
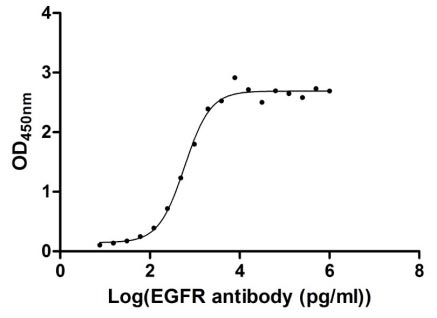
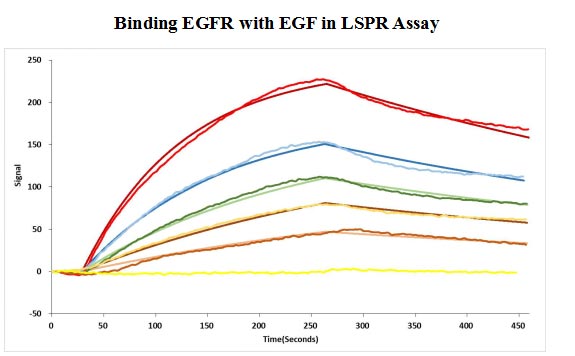
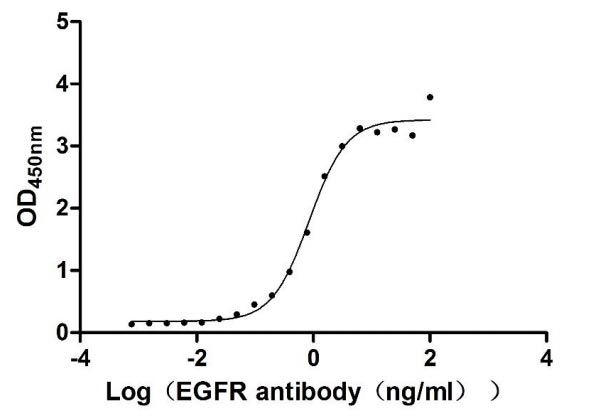
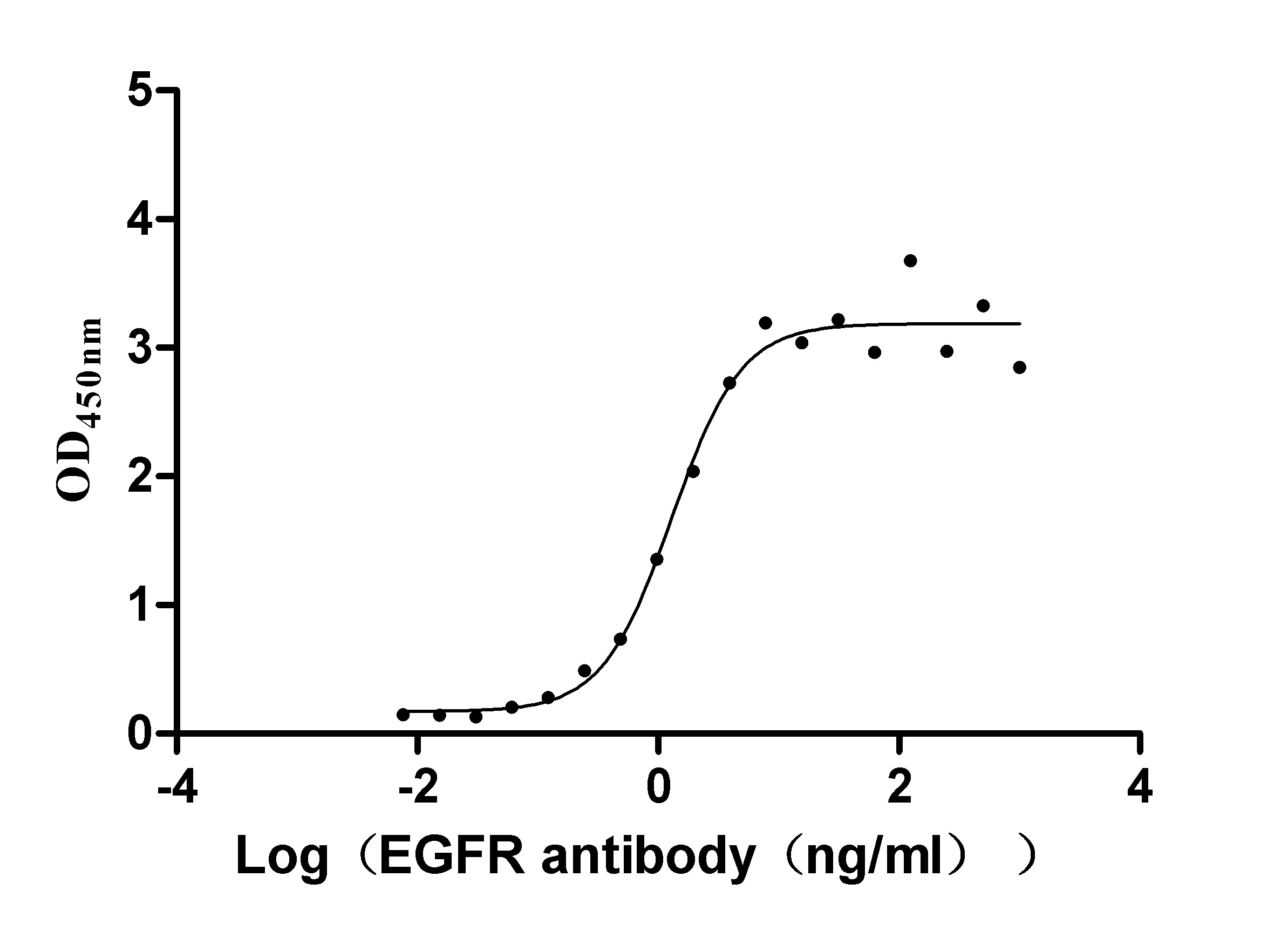
The gene coding for the human Epidermal growth factor receptor (EGFR) protein (25-645aa) is co-introduced into a plasmid vector with the N-terminal 10xHis-tag gene and the C-terminal Myc-tag gene, creating recombinant plasmid that is subsequently transformed into mammalian cells. mammalian cells capable of surviving in the presence of a specific antibiotic are selected and then cultured under conditions favorable for the expression of the gene of interest. Post-expression, the recombinant human EGFR protein is isolated and purified from the cell lysate using affinity purification. Denaturing SDS-PAGE is applied to resolve the resulting recombinant human EGFR protein, revealing a purity exceeding 90%. This recombinant human EGFR protein has been validated as active in functional ELISA and LSPR assy. Its endotoxin content is less than 1.0 EU/ug as determined by the LAL method.
The Epidermal Growth Factor Receptor (EGFR) is a protein on cell surfaces that guides vital cell activities. It gets activated when specific molecules, like EGF, bind to it, setting off a chain reaction inside cells [1]. When EGFR doesn't work properly, it's closely tied to various cancers, including brain, lung, and breast cancer [2]. Recent research shows EGFR can also help cancer cells survive even without its typical signaling, suggesting it is a potential target for new cancer treatments [3][4]. Moreover, EGFR sparks important cell pathways, controlling how cells respond to their environment [5].
In cancer, EGFR seems to help lung tumors dodge the immune system by ramping up substances that dampen immune responses [6]. It's also linked to controlling a protein called PD-L1 in esophageal cancer, influencing how the immune system reacts [7]. Plus, EGFR kicks off a process called nuclear factor κB signaling, which fuels the growth of EGFR-related tumors [2]. Understanding how lung cancer patients respond to drugs targeting EGFR is tricky, showing the challenges in treating EGFR-related cancers [8].
EGFR isn't just about cancer—it's involved in many cell jobs like cell death, growth, and specialization through its signals [9][10]. It even plays a role in the growth and movement of cells important for pregnancy, showing its relevance beyond cancer [11]. Additionally, EGFR is connected to activating a protein called cyclooxygenase-2, which drives certain cancers forward [12].
References:
[1] M. Iwamoto, W. Saso, R. Sugiyama, K. Ishii, M. Ohki, S. Nagamoriet al., "Epidermal growth factor receptor is a host-entry cofactor triggering hepatitis b virus internalization", Proceedings of the National Academy of Sciences, vol. 116, no. 17, p. 8487-8492, 2019. https://doi.org/10.1073/pnas.1811064116
[2] D. Pan and X. Lin, "Epithelial growth factor receptor–activated nuclear factor κb signaling and its role in epithelial growth factor receptor–associated tumors", The Cancer Journal, vol. 19, no. 6, p. 461-467, 2013. https://doi.org/10.1097/ppo.0000000000000001
[3] R. Thomas and W. Zhang, "Rethink of egfr in cancer with its kinase independent function on board", Frontiers in Oncology, vol. 9, 2019. https://doi.org/10.3389/fonc.2019.00800
[4] R. Katreddy, L. Bollu, F. Su, N. Xian, S. Srivastava, R. Thomaset al., "Targeted reduction of the egfr protein, but not inhibition of its kinase activity, induces mitophagy and death of cancer cells through activation of mtorc2 and akt", Oncogenesis, vol. 7, no. 1, 2018. https://doi.org/10.1038/s41389-017-0021-7
[5] M. Hiroshima, M. Abe, N. Tomishige, F. Hullin-Matsuda, A. Makino, M. Uedaet al., "Membrane cholesterol interferes with tyrosine phosphorylation but facilitates the clustering and signal transduction of egfr",, 2021. https://doi.org/10.1101/2021.08.28.457965
[6] E. Akbay, S. Koyama, J. Carretero, A. Altabef, J. Tchaicha, C. Christensenet al., "Activation of the pd-1 pathway contributes to immune escape in egfr-driven lung tumors", Cancer Discovery, vol. 3, no. 12, p. 1355-1363, 2013. https://doi.org/10.1158/2159-8290.cd-13-0310
[7] W. Zhang, Q. Pang, C. Yan, Q. Wang, J. Yang, S. Yuet al., "Induction of pd-l1 expression by epidermal growth factor receptor–mediated signaling in esophageal squamous cell carcinoma", Oncotargets and Therapy, vol. Volume 10, p. 763-771, 2017. https://doi.org/10.2147/ott.s118982
[8] Z. Liang, J. Zhang, X. Zhang, J. Gao, S. Wu, & T. Liu, "Relationship between egfr expression, copy number and mutation in lung adenocarcinomas", BMC Cancer, vol. 10, no. 1, 2010. https://doi.org/10.1186/1471-2407-10-376
[9] T. Matsuo, S. Nishizuka, K. Ishida, T. Iwaya, M. Ikeda, & G. Wakabayashi, "Analysis of the anti-tumor effect of cetuximab using protein kinetics and mouse xenograft models", BMC Research Notes, vol. 4, no. 1, 2011. https://doi.org/10.1186/1756-0500-4-140
[10] T. Yoshida, I. Okamoto, T. Okabe, T. Iwasa, T. Satoh, K. Nishioet al., "Matuzumab and cetuximab activate the epidermal growth factor receptor but fail to trigger downstream signaling by akt or erk", International Journal of Cancer, vol. 122, no. 7, p. 1530-1538, 2008. https://doi.org/10.1002/ijc.23253
[11] X. Bai, C. Zheng, & N. Ren, "Trophoblast proliferation, migration, and invasion is regulated by the mir-195/egfr signaling pathway in recurrent spontaneous abortion patients",, 2019. https://doi.org/10.1101/630590
[12] H. Lo, X. Cao, H. Zhu, & F. Ali‐Osman, "Cyclooxygenase-2 is a novel transcriptional target of the nuclear egfr-stat3 and egfrviii-stat3 signaling axes", Molecular Cancer Research, vol. 8, no. 2, p. 232-245, 2010. https://doi.org/10.1158/1541-7786.mcr-09-0391