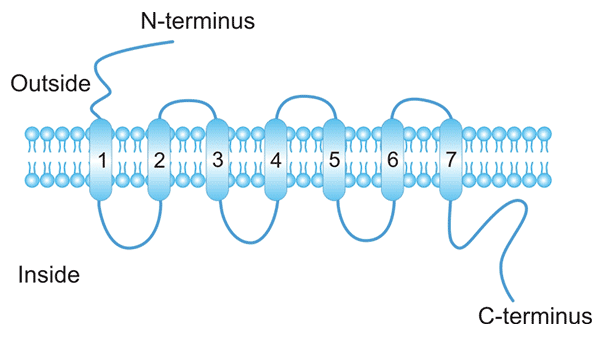
[1] Samantha J. Allen, Susan E. Crown, et al. Chemokine: Receptor Structure, Interactions, and Antagonism [J]. Annu. Rev. Immunol. 2007. 25:787–820.
[2] Graham GJ, McKimmie CS. Chemokine scavenging by D6: a movable feast [J]? Trends Immunol. 2006, 27:381–86.
[3] Weber M, Blair E, et al. The chemokine receptor D6 constitutively traffics to and from the cell surface to internalize and degrade chemokines [J]. Mol. Biol. Cell. 2004, 15:2492–508.
[4] Nibbs R, Graham G, et al. Chemokines on the move: control by the chemokine“interceptors” Duffy blood group antigen and D6 [J]. Semin. Immunol. 2003, 15:287–94.
[5] Lefkowitz RJ. Historical review: a brief history and personal retrospective of seventransmembrane receptors [J]. Trends Pharmacol. Sci. 2004, 25:413–22.
[6] Murdoch.Craig, Finn. Adam. Chemokine receptors and their role in inflammation and infectious diseases [J]. Blood. 2000, 95: 3032–3043.
[7] Annelien J.M. Zweemer, Jimita Toraskar, et al. Bias in chemokine receptor signaling [J]. Trends in Immunology. 2014, 35 (6):243-252.
[8] MukaidaN, BabaT. Chemokines in tumor development and progression [J]. Exp CellRes. 2012, 318(2):95–102.
[10] MariaVela, Mariana Aris, et al. Chemokine receptor-specific antibodies in cancer immunotherapy: achievements and challenges [J]. 2015, 6 (12): 1-15.
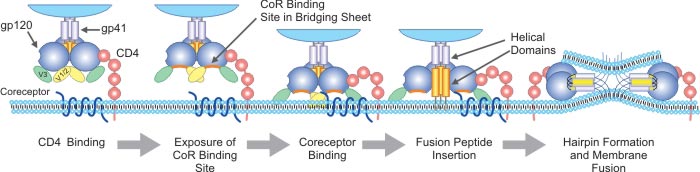
[11] Katrien Princen, Dominique Schols. HIV chemokine receptor inhibitors as novel anti-HIV drugs [J]. Cytokine & Growth Factor Reviews. 2005, 16: 659–677.
[12] Broder CC, Collman RG. Chemokine receptors and HIV [J]. J Leukoc Biol. 1997, 62(1):20-9.
[13] Doms RW. Beyond receptor expression: the influence of receptor conformation, density, and affinity in HIV-1 infection [J]. Virology. 2000, 276:229–37.
[14] RotA, von Andrian UH. Chemokines in innate and adaptive host defense: basic chemokinese grammar for immune cells [J]. Annu Rev Immunol. 2004, 22: 891–928.
[15] WhiteGE, IqbalAJ,et al.CC chemokine receptors and chronic inflammation therapeutic opportunities and pharmacological challenges [J]. Pharmacol Rev. 2013, 65(1):47–89.
[16] BarbieriF, BajettoA, et al. Role of chemokine network in the development and progression of ovarian cancer: a potential novel pharmacological target [J]. J Oncol. 2010, 42: 56-69.
[17] Balk will F. Chemokine biology in cancer [J]. Semin Immunol. 2003, 15(1):49–55.
[18] Viola A, Sarukhan A, et al. The pros and cons of chemokines in tumor immunology [J]. Trend sImmunol. 2012, 33(10):496–504.
[19] Burger JA, Kipps TJ. CXCR4: a key receptor in the crosstalk between tumor cells and their micro environment [J]. Blood. 2006, 107(5):1761–7.
[20] Balkwill F. The significance of cancer cell expression of the chemokine receptor CXCR4 [J]. Semin Cancer Biol. 2004, 14(3):171–9.