After the rise of recombinant DNA technology, scientists faced an important problem: how to purify the expressed recombinant proteins efficiently. Traditional protein purification methods are often cumbersome and inefficient and require developing a specific purification strategy for each protein.
Immobilized metal ion affinity chromatography (IMAC) was first proposed in 1975 [1,2]. In 1987, Hochuli and his colleagues first described a general purification method for the recombinant protein tagged with a poly-histidine peptide through IMAC [3,4]. The rapid development of IMAC technology has made the use of his tags more common.
This article mainly introduces the definition, functions, and applications of His tags, as well as some frequently asked questions (FAQs) about the purification of His-tag fusion protein.
Contents
1.What Is A His Tag?
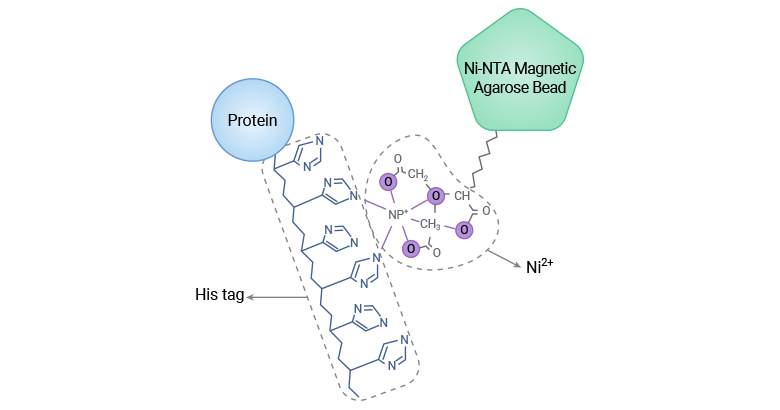
A His tag, also called polyhistidine tag, is typically comprised of a string of 6-10 consecutive histidine residues, each of which has an imidazole ring that can form stable coordination bonds with transition metal ions such as Ni2+ and Co2+ to selectively combine with metal ions, the basis for his-tagged protein purification [5].
Figure 1. The mechanism of His-tagged protein purification through IMAC
The molecular weight of His tags changes depending on the number of histidine residues incorporated into the tag. The hexahistidine (6xHis)-tag is the smallest tag with a molecular weight of about 0.84 kDa and is the most common affinity tag used for protein purification.
His-tagging is the process in which a His tag (selected number of histidines) is added to either the N- or C-terminus of the targeted recombinant protein. The placement of the His tag is specific to each protein and is determined by many factors, including the protein’s function and structure, expression system and expression level, ease of purification and detection, and post-translational modifications.
2. What Are Functions of His Tags?
The addition of His-tags to the protein of interest can form epitopes and unique structural features ( for ligand binding) that conducive to protein detection and purification, respectively.
2.1 Protein Purification
Polyhistidine tags allow for facile protein purification using immobilized metal affinity chromatography (IMAC) [5]. His-tagged proteins can be eluted using high concentrations of imidazole, which competes with His tags for binding sites to metal ions, thereby dissociating the target protein from the affinity column. High concentrations of imidazole usually do not affect protein structure and function.
His-tag, as the preferred tag for protein purification, has the following advantages:
a.N-terminal His-Tag is compatible with the transcription and translation mechanism of bacteria, which is conducive to protein expression;
b.Purification of His-Tag fusion protein based on IMAC is more convenient;
c.His-tag has little effect on the properties of the target protein and does not change the solubility and biological function of the target protein;
d.The immunogenicity of His-Tag is relatively low, and the purified His-tagged protein can be directly injected into the animal for immunization and preparation of antibodies;
e.Multiple affinity tag domains have been successfully combined with the polyhistidine tag on recombinant proteins, including the GST tag [6], a modified S-peptide of ribonuclease A [7], and both the albumin-binding protein fusion domain and a biotinylation accepting domain [8].
2.2 Protein Detection
Anti-His antibodies can specifically bind to His-tagged proteins, enabling their detection and quantification on Western blots [9].
His-tagged proteins can be detected and quantified using anti-His antibodies in ELISA assays.
In pull-down assay, His tagged proteins are immobilized on the affinity resin containing nickel or cobalt ions, enabling the capture and analysis of the binding proteins.
2.3 Protein Localization
Fluorescently labeled anti-His antibodies allow for the visualization of His-tagged proteins within cells using immunofluorescence microscopy, facilitating studies of protein localization and expression.
3. Applications of His Tags
As an important biotechnology tool, His tag plays an important role in protein purification, detection, and engineering, and is widely used in scientific research and the biological industry.
3.1 Recombinant Protein Production
The recombinant proteins can be co-expressed with His tags by adding the sequence encoding His tag into the expression vector of the target protein. His tags can help proteins correctly fold and improve their expression and purity. His tags also increase the solubility of the expressed proteins.
The resulting His-tagged recombinant proteins can be purified efficiently through IMAC. IMAC purification method is commonly used for the purification of many His-tagged recombinant proteins expressed in bacterial, yeast, insect, and mammalian expression systems. This method enables rapid and efficient purification of target proteins from complex mixtures and is suitable for large-scale protein purification.
The popularity of IMAC purification is attributed in part to its numerous advantageous features such as the strong affinity of the His-tag in recombinant proteins for nickel-nitrilotriacetic agarose (Ni-NTA) beads and the easy elution process with imidazole buffer.
3.2 Biological Research
His tag fusion proteins can also be used to study protein-protein interactions, protein-DNA interactions, as well as protein structure and function studies.
The high purity of His-tagged proteins obtained through IMAC purification is crucial for structural studies, including X-ray crystallography and nuclear magnetic resonance (NMR) spectroscopy. These techniques require homogeneous protein samples to determine the three-dimensional structures of proteins. His tags usually do not affect the crystallization process and can even participate in the formation of crystal contacts.
His-tagged proteins can also be used in several experimental assays to identify and characterize protein-protein interactions. These assays include WB, ELISA, pull-down assay, immunoprecipitation (IP), surface plasmon resonance (SPR), and co-immunofluorescence.
Additionally, His-tagged proteins can be used in various functional assays to study enzyme kinetics, substrate specificity, and inhibition. The high purity of the proteins ensures accurate and reproducible results.
His tag can also be used to detect the presence and quantification of target proteins. By using antibodies or nickel ion dyes with His tag, proteins with His tag can be specifically detected and quantified, which is very helpful for studying protein expression and localization.
4. Related FAQs about Purification of His-tag Fusion Protein
During the purification of His-tagged proteins, some common problems may be encountered. The following is a detailed description of these problems and their corresponding solutions:
① The target protein is expressed insufficiently, resulting in low yield during purification.
Solution:
- Optimize expression conditions: adjust the inducer concentration, induction time, temperature, and other conditions. For example, lowering the temperature can increase the solubility of the protein.
- Use different host strains: select strains with more efficient expression, such as BL21(DE3), Rosetta, etc.
- Gene optimization: optimize the codon of the target gene to improve the expression efficiency in the host.
② The target protein forms insoluble inclusion bodies during expression.
Solution:
- Lower the expression temperature: lower the expression temperature to 16-25°C, slow down the expression rate, and promote correct folding.
- Use denaturation and renaturation methods: dissolve the inclusion bodies under denaturing conditions (such as 6M urea or 8M guanidine hydrochloride), and then renature the protein by dialysis or dilution.
- Add co-factors or denaturants: add cofactors (such as glycerol, ammonium sulfite) or denaturants (such as amino acids, sugars) to the culture medium to help the protein fold correctly.
③ Other proteins bind non-specifically to Ni-NTA or Co-NTA resins, resulting in more impurities in the purified samples.
Solution:
- Optimize washing conditions: increase the imidazole concentration in the wash buffer (e.g., 20-40mM) to remove non-specific binding proteins.
- Use high-salt buffer: increase the salt concentration (e.g., 500mM NaCl) in the wash buffer to reduce non-specific binding.
- Increase the number of washes: further remove impurities by increasing the number of washes.
④ His-tagged proteins are degraded by proteases during expression or purification.
Solution:
- Use protease inhibitors: add protease inhibitor cocktails (e.g., PMSF, EDTA, leupeptin, etc.) during lysis and purification.
- Lower the temperature: keep the temperature low (e.g., 4°C) during lysis and purification to reduce protease activity.
- Select a degradation-resistant host strain: use a strain lacking a specific protease such as BL21(DE3)pLysS.
⑤ His-tagged proteins cannot bind to Ni-NTA or Co-NTA resins effectively.
Solution:
- Check the integrity of the His tag: make sure that the His tag is not removed or degraded by the enzyme.
- Adjust the pH: make sure the pH of the binding buffer is around 8.0, because the His tag can effectively bind metal ions at a higher pH.
- Reduce EDTA or other chelating agents: make sure that the lysis and purification buffers do not contain chelating agents (such as EDTA) to avoid chelating metal ions and affecting binding.
⑥ Protein inactivation during purification.
Solution:
- Optimize purification conditions: make sure that the buffer and environmental conditions during purification are suitable for protein stability (such as pH, ionic strength, and temperature).
- Add protective agents: add protective agents (such as glycerol, DTT) to the purification buffer to prevent protein denaturation or oxidation.
- Use or store as soon as possible: try to shorten the purification time, perform activity assays immediately after purification or store under appropriate conditions (such as low temperature, appropriate buffer).
⑦ Low elution efficiency of target protein, resulting in low yield.
Solution:
- Increase imidazole concentration: increase the imidazole concentration in the elution buffer (such as 250-500mM) to effectively elute the His-tagged protein.
- Optimize the composition of the elution buffer: adjust the pH and salt concentration of the elution buffer to improve the elution efficiency.
- Extend the elution time: increase the elution time or elution volume to ensure that the target protein is completely eluted.
⑧ The protein is unstable after elution and is prone to aggregation or degradation.
Solution:
- Add stabilizers: add stabilizers (such as 20% glycerol, 1mM DTT) to the elution buffer to improve the stability of the protein.
- Use a suitable buffer: choose a buffer that is better for the stability of the target protein (such as Tris, Hepes).
- Quick processing and storage: perform subsequent steps (such as dialysis, concentration) as quickly as possible and store under appropriate conditions (such as -80°C).
Some common problems encountered in the purification of His-tagged proteins can be effectively solved by optimizing these conditions and taking appropriate measures, enabling efficient purification of His-tagged proteins and providing high-quality protein samples for subsequent functional research and applications.
Conclusion
His tag can specifically bind to Ni-NTA or Co-NTA resin, greatly simplifying the purification process of His-tagged proteins and improving purification efficiency and yield. Furthermore, His-tag antibodies can detect and locate His-tagged proteins, which makes His tags widely used in protein-protein interaction research, enzyme activity research, protein structural biology, ligand interaction research, and other fields and become an indispensable tool in molecular biology and biochemistry experiments.
CUSABIO can provide several His-tag antibodies helpful for the detection of His-tagged recombinant proteins in relevant research.
References
[1] Porath N, Ben-Shaul Y, Friedberg I. Proceedings: Some properties of membrane-bound adenosine triphosphatase of Euglena gracilis chloroplasts. Isr J Med Sci. 1975;11:1189–1190.
[2] Porath J, Carlsson J, et al. (1975) Metal chelate affinity chromatography, a new approach to protein fractionation [J]. Nature 258: 598–599.
[3] Hochuli E, Dobeli H, Schacher A. New metal chelate adsorbent selective for proteins and peptides containing neighbouring histidine residues. J Chromatogr. 1987;411:177–184.
[4] Hochuli E. Large-scale chromatography of recombinant proteins. J Chromatogr. 1988;444:293–302.
[5] Riguero V, Clifford R, et al. Immobilized metal affinity chromatography optimization for poly-histidine tagged proteins [J]. J Chromatogr A. 2020 Oct 11;1629:461505.
[6] Panagiotidis CA, Silverstein SJ. pALEX, a dual-tag prokaryotic expression vector for the purification of full-length proteins [J]. Gene. 1995 Oct 16;164(1):45-7.
[7] Kim JS, Raines RT. Peptide tags for a dual affinity fusion system [J]. Anal Biochem. 1994 May 15;219(1):165-6.
[8] Nilsson J, Larsson M, et al. Multiple affinity domains for the detection, purification and immobilization of recombinant proteins [J]. J Mol Recognit. 1996 Sep-Dec;9(5-6):585-94.
[9] Zentgraf H, Frey M, et al. Detection of histidine-tagged fusion proteins by using a high-specific mouse monoclonal anti-histidine tag antibody [J]. Nucleic Acids Res. 1995;23:3347–3348.





Comments
Leave a Comment